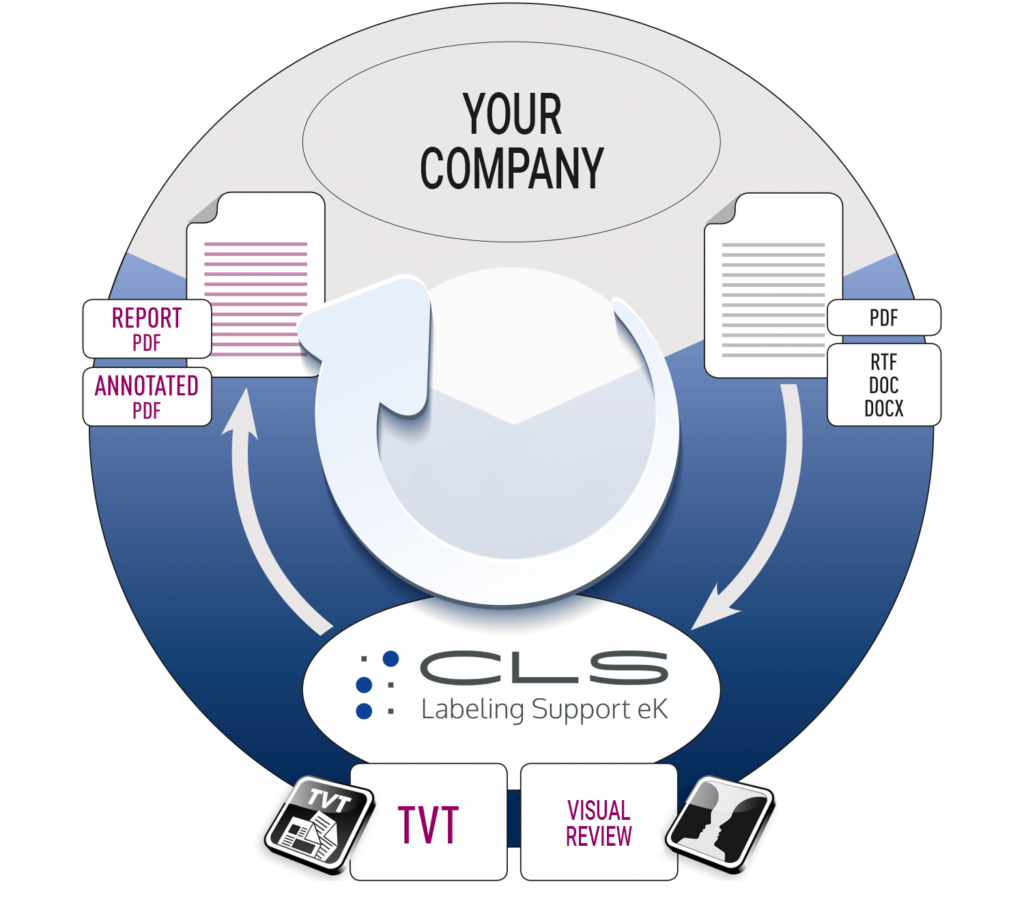
As in all areas related with publication of texts, the pharmaceutical industry also requires text review and approval.
However, the text comparison in the pharmaceutical industry consists not only of a one-to-one review of the manuscript and the final printable PDF, but also of recognizing logical correlations when evaluating the deviations. This includes, for example, text and figure references as well as the correct registration office for side effects.
What else to be considered!
Typographical errors after approval
Sometimes, minor typing errors are only detected during critical proofreading shortly before approval for printing and, depending on the approval procedure, can often not be corrected that easily. In the centralized procedure, typos are basically officially approved.
Tight Timelines
Tight timelines in the production facilities tempt you to read quickly or carelessly, especially if the content of the text is already known or even created by yourself. This could pose the risk of errors not being recognized, or a mere electronic text comparison being relied on.
Having the print version reviewed independently of the text owner before the final approval is therefore an important part of quality assurance.
Review & Support
For that reason, we offer a close cooperation with simple processes and support in your review responsibility by: